Vol. 1, Issue 3 (2013)
Studies of Some Physicochemical Properties of Isatin
![The plot of pH against volume of NaOH in [A] methanol + water and [B] 1,4-dioxane + water at 298.15K.](https://www.chemijournal.com/up/graphics/30/19.1.jpg)
Fig. 1: The plot of pH against volume of NaOH in [A] methanol + water and [B] 1,4-dioxane + water at 298.15K.
![The variation of velocity (U) with concentration for Isatin in [A] methanol and [B] 1,4-dioxane.](https://www.chemijournal.com/up/graphics/30/19.2.jpg)
Fig. 2: The variation of velocity (U) with concentration for Isatin in [A] methanol and [B] 1,4-dioxane.
![The variation of intermolecular free path length (Lf) against concentration for Isatin in [A] Methanol and [B] 1,4-dioxane.](https://www.chemijournal.com/up/graphics/30/19.3.jpg)
Fig. 3: The variation of intermolecular free path length (Lf) against concentration for Isatin in [A] Methanol and [B] 1,4-dioxane.
![The variation of adiabatic compressibility (κs) against concentration for Isatin in [A] Methanol and [B] 1,4-dioxane](https://www.chemijournal.com/up/graphics/30/19.4.jpg)
Fig. 4: The variation of adiabatic compressibility (κs) against concentration for Isatin in [A] Methanol and [B] 1,4-dioxane
![The variation of adiabatic compressibility (κs) against concentration for Isatin in [A] Methanol and [B] 1,4-dioxane](https://www.chemijournal.com/up/graphics/30/19.5.jpg)
Fig. 5: The variation of adiabatic compressibility (κs) against concentration for Isatin in [A] Methanol and [B] 1,4-dioxane
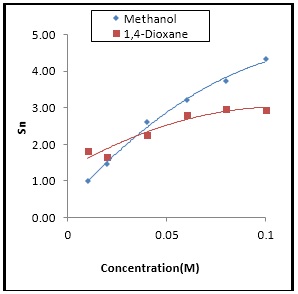
Fig. 6: The variation of Sn against concentration for Isatin in Methanol and 1,4-dioxane




