Vol. 1, Issue 3 (2013)
Study on Complex Formation of Cu(II) with Water Insoluble PAN in Acidic Water, Using UV-Vis Spectrophotometer
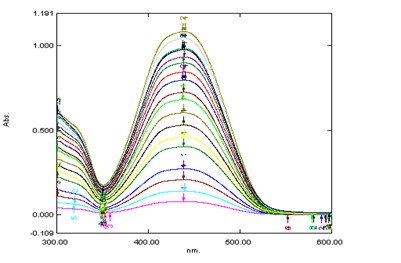
Fig. 1: (a). Absorption spectrum at different concentration of PAN (4.0×10-6M/L to 8.0×10-5M/L) at pH=2.5, λMax=440 nm in aqueous solution.
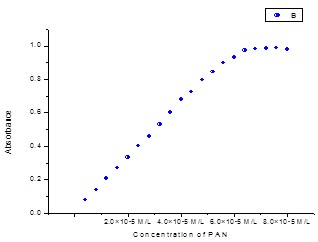
Fig. 2: (b). Absorption of different concentration of PAN (4.0×10-6M/L to 8.0×10-5M/L) at pH=2.5, λMax=440 nm in aqueous solution.
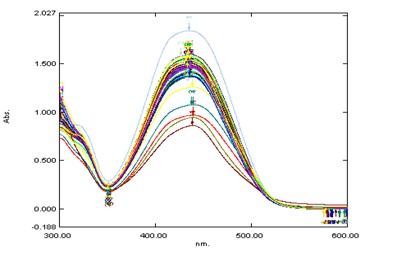
Fig. 3: (c). Absorption spectrum at different pH (1.00 - 3.00) of PAN (6.0×10-6M/L) at λMax=440nm in aqueous solution.
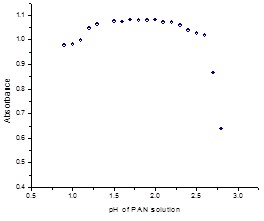
Fig. 4: (d). Absorbance at different pH (1.00 --3.00) of PAN (6.0×10-6M/L) at λMax=440nm in aqueous solution.
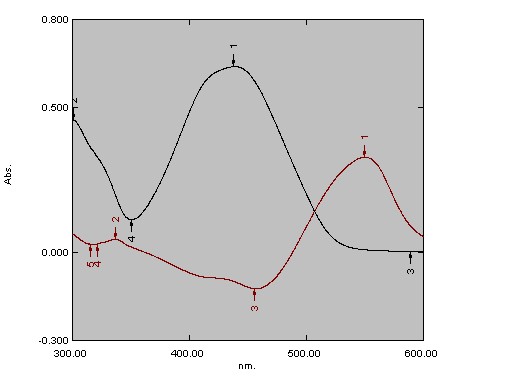
Fig. 5: Absorption spectrum of PAN and Cu(II)-PAN against the reagent blank at pH=2.50 in aqueous solution.
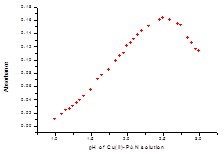
Fig. 6: Effect of pH on the absorbance of Cu(II)-PAN 1:10) complex.
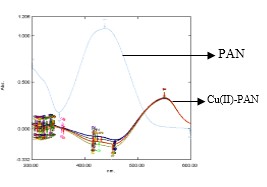
Fig. 7: The overlay of absorption spectrum of Cu(II)-PAN (1:10) at different time.
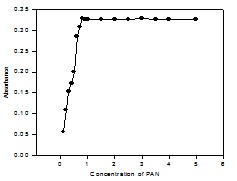
Fig. 8: Effect of reagent (PAN) molar concentration ratio on the absorbance of Cu(II)-PAN system.




